Mauro Usability Science
In Human Factors Research & Usability Testing Consulting for Leading Corporations, Startups, NGO and Government Agencies Since 1975
Divider 01
We offer a proven history of solving complex human factors engineering and usability problems for the world’s leading corporations, startups and government agencies.
Divider 02

We solve usability and human factors challenges through rigorous, science-based methods grounded in decades of experience.
Our success comes from blending deep knowledge of the Human Operating System (hOS) with the complexities of advanced technology (tOS). Our methodology ensures both business and user needs are addressed with precision.
We are fluent in all critical usability research methods and pioneered the use of several of these methods in the U.S. in the early 1970s. In all formative and summative projects, we employ the appropriate methods required to answer your complex business questions.
Our neuroscience-based advanced data capture methodologies deliver robust quantitative data to answer client’s most complex medical device research questions, employing methodologies such as 3D Spatial Tracking, Newtonian Force Measurement, Electromyography (EMG), Eye-Tracking, Micro-Facial Expression Analysis (Emotional Response), and others.
We have extensive expertise with critical user populations, ranging from COPPA age children to individuals of advanced age with severe medical conditions, as well as complex healthcare provider (HCP) and patient populations.
We specialize in highly focused and experienced usability testing and human factors engineering optimization expertise for business-critical, time-sensitive FDA responses.
Our heuristics analyses provide immediate feedback on critical usability and user experience issues present in your medical product or system. We've conducted extensive heuristics on complex medical products, drug delivery devices, imaging systems, patient EMRs and related processes using specialized heuristics rule sets directed at FDA HE 75 guidelines.
We have extensive experience in the design and testing of medical product instructions for use (IFU) and reference guide (RG) systems. We work with clients to ensure that FDA guidance and mandated human factors engineering performance criteria are met for IFU and RG components of medical and drug delivery devices.
Divider 03
Our firm has executed over 3,000 human factors and usability testing projects ranging over a period of 50 years. This background offers our clients the benefit of knowledge transfer across verticals and within their specific sector.


Medical Products and Systems
01In the medical products space we offer clients an exceptionally deep and broad level of experience focusing on FDA HF guidance and usability testing. We have developed, validated and utilized in business-critical medical device programs the most advanced testing methods in industry. We are experts in the application of FDA, ISO and other human factors optimization issues.


Consumer / Commercial / Industrial Products & Systems
02We have developed deep industry experience solving all manner of business-critical HF and UT problems. No other consulting firm in our category has more experience. Click here to visit our extensive content on consumer, commercial, industrial and military systems human factors and usability testing.



Institute for Design Science and Public Policy
03We established the Institute for Design Science and Public Policy (IDSPP) to provide a professional framework for expanding the field of design science and public policy with the specific mission of advancing applied design research and design IP protections through professional and governmental involvement.
Citations & awards
We’ve received more professional citations and awards than any other HF / UT consulting firm including reception of the 2024 Titan of Industry For Human Factors Research Award and citations from NASA, Association for Computing Machinery, Human Factors and Ergonomics Society, Industrial Designers Society of America and even an Emmy award for virtual world research.
Recipient of the 2024 Titan of Industry For Human Factors Research Award




Divider 04
Our case study archive reflects the breadth and depth of our experience.
New York Stock Exchange

United States Department of Defense
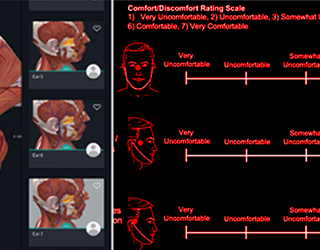
Augmented Reality (AR) Glasses Comfort/Fit and Feature Set Validation and Definition Research Studies

Applied Cognitive Task Analysis (ACTA) for Complex Thoracolumbar Decompression and Fusion Spine Procedure Enabling Technology
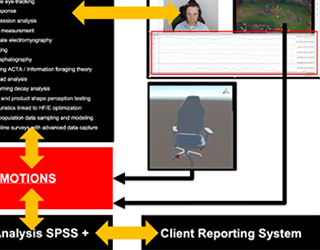
Neuroscience-Based Human Factors and Ergonomics Testing Platform for Video Game Systems and Workstations

PFS Force Specification Study with Off-Axial Force Measurement
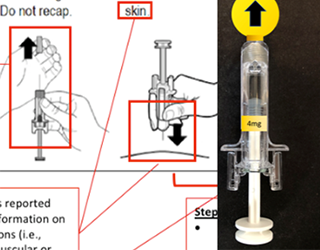
Opioid Overdose Drug Delivery Study
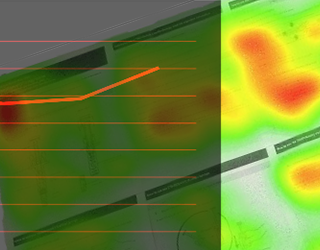
Agile Medical Device Summative Human Factors/Usability Testing Focusing on Rapid IFU Optimization

Intravitreal Injection Pre-filled Syringe Optimization Studies

Major Pharmaceutical Clients
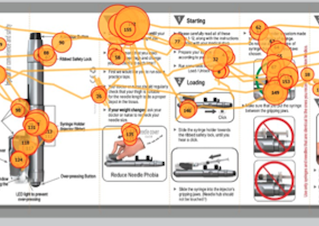
High Resolution Eye-Tracking for Medical Device System Optimization

Citibank
Harvard Endowment

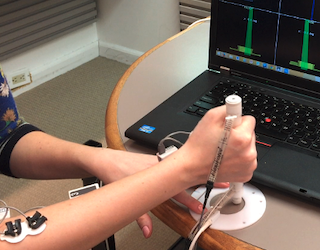
Drug Injector Newtonian Force Measurement

NASA

Whitney Museum of Art
Drug Injector 3D Spatial tracking

Tiffany & Company

Google ATAP

Apple
MTV Viacom

Sony

Leukemia and Lymphoma Society LLS
Divider 05
Our publications demonstrate our established position as a thought leader in the field.
-
Medical device usability testing: 38 important guidance documents and research papers for medical device development
-

A critical analysis of FDA guidance for user percentile device design criteria versus currently available human factors engineering data sources and industry best practices
-

Analyzing current FDA human factors IFU guidance through the lens of a rapidly changing medical drug delivery device landscape
-

User Interface Design and UX Design: 80+ Important Research Papers Covering Peer-Reviewed and Informal Studies
-

Development and Initial Validation of an Empirical Ordinary Observer Test for Design Patent Infringement
-

Apple v. Samsung Amicus Brief: Brief of amici curiae brief 113 distinguished industrial design professionals and educators in support of respondent
-

GM v. KSP En Banc Federal Circuit Appeal Amicus Brief: Corrected Brief of Amici Curiae Institute for Design Science and Public Policy and Thirty-Six Distinguished Industrial Designers in Support of Neither Party
-

Columbia Sportswear v. Seirus Federal Circuit Appeal Amicus Brief: Brief of Amicus Curiae Industrial Designers Society of America, Inc.’s In Support of the Petition for Rehearing and Rehearing En Banc
-

Why the Future of Design Patent Protections Will Rely on Modern Neuroscience, Not Constitutional and Legal Reversionism
-
Development and validation of neuroscience-based system human factors and ergonomic assessment methodologies for measurement of impact of seating system design on video gameplay performance
-

Understanding costs and risks for HFE usability studies – Part 1: Testing In-House
-

Challenges implementing new advanced HFE testing methods
Divider 06
Testimonial

Whether you're facing usability issues, compliance risks, or technology adoption hurdles, our team is ready to help. Let’s work together to create solutions that actually work—for your business and your users.